-
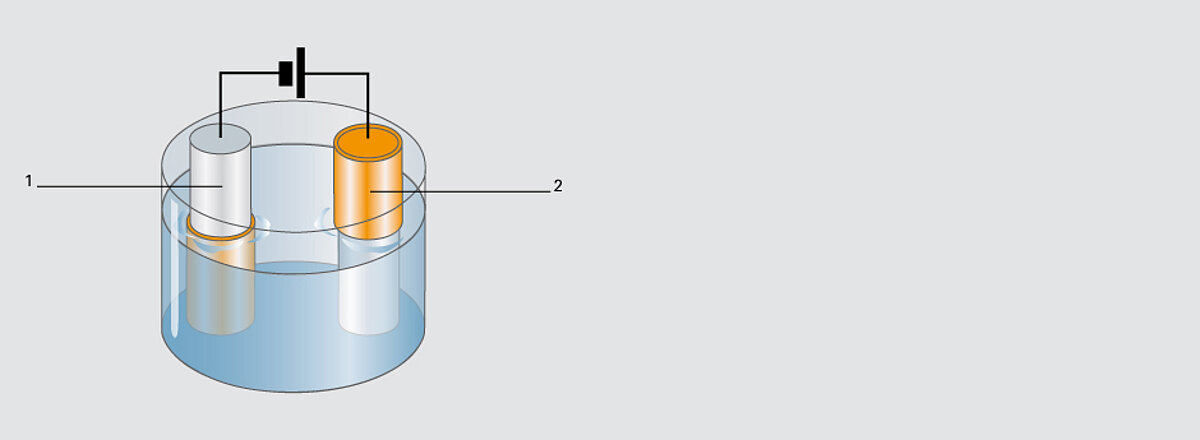
Electroplating process
The electroplating process is used to apply required metallic layers required for imaging or refinement on the steelbase. For this the cylinder comes into contact with a metallic salt solution (electrolytic bath) and is liked to the minus pole (cathode). If electricity is fed to the bath, an ion migration occurs in keeping with the electrochemical principle. The metal located at the plus pole (anode) (copper, nickel, chrome or zinc) is attracted from the cylinder to the minus pole. An equivalent metal layer is created on the cylinder. Depending on the starting material and desired final product, certain electrolytes are used and the layers applied or processed in a certain sequence. By using different galvanic processes, cylinders can be copper-coated, zinc-coated, chromed and nickel-plated.